Think back to March 2020. The Centers for Disease Control and Prevention (CDC) was just beginning to see a decline in flu activity and there was even chatter that the annual flu season was coming to a slow but steady close.
Then BOOM. The SARS-CoV-2 strain swooped in and stole the show. On March 11, the World Health Organization (WHO) declared COVID-19 a pandemic, two days later the United Stated was officially in a national emergency, and the flu pretty much fell off everyone’s radar.
Here’s the thing. The flu never left. And if the COVID-19 pandemic taught us anything, it’s that a novel and life-threatening virus strain can come out of nowhere. Put the two facts together and it’s entirely possible that next season – or the season some time after that – could bring a new and deadly influenza strain that spreads between people at a dizzying pace.
A vaccine could veil a deadly influenza virus strain and keep it from achieving pandemic proportions – just as a vaccine is expected to end the COVID-19 nightmare – but to really make a difference, it would need to be a long-lasting universal vaccine that induces immune responses to a wide spectrum of influenza virus strains and subtypes.
A universal influenza vaccine isn’t a new idea, but headway has been slow – until recently. A team of researchers at the Icahn School of Medicine at Mount Sinai recently presented “strong and durable results” in a Phase 1 clinical trial for a vaccine that produces antibodies that target the part of the surface protein of the influenza virus known to neutralize diverse influenza strains and therefore induce immune responses to a wide spectrum of influenza virus strains and subtypes. The vaccine, thus far evaluated for safety and effectiveness in 65 participants, delivered an immune response that endured for at least 18 months following vaccination.1
Study results, published in the journal Nature Medicine, November 2020, explains in detail how this chimeric hemagglutinin (HA)-based vaccine has the potential to provide long-lived protection with two or three immunizations. If achieved, results would significantly reduce the likelihood of an emerging virus subtype or strain that can push beyond the vaccine’s wall of protection and potentially trigger an influenza pandemic.
The Same But Different
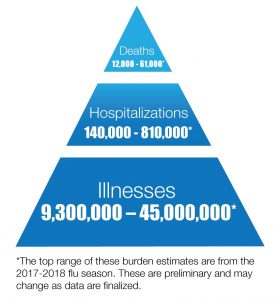
While different viruses cause influenza and COVID-19, history confirms that the two infections are equally serious. Since 2010, the Centers for Disease Control and Prevention (CDC) reports that influenza has annually brought between 9 million to 45 million illnesses, 140,000 to 810,000 hospitalizations, and 12,000 to 61,000 deaths since 2010. As for pandemic level statistics, the CDC estimates that about 500 million people, or one-third of the world’s population, became infected with the 1918 H1N1 influenza virus, with the number of deaths was estimated to be at least 50 million worldwide and about 675,000 in the United States.2, 3
Anybody’s Guess
The world does, of course, have seasonal influenza vaccines, with each year’s new model containing three or four influenza virus strains based on global health surveillance and predictions. However, if educated guesses don’t exactly match the viruses actually circulating, vaccine results are underwhelming. For example, the CDC estimated that the 2018-2019 flu vaccine was only 29% effective after a brutal flu strain that season blind-sided everyone. The CDC’s highest estimated seasonal influenza vaccine effectiveness score came in at 60% effective and went to the 2010-2011 vaccine.4
Th Mount Sinai researchers’ chimeric HA vaccine works around guesstimates by targeting a different part of the hemagglutinin protein, the major surface glycoprotein of the influenza virus that binds to host cell receptors. As outlined in the study’s published paper, conventional vaccines induce neutralizing antibodies that target the distal part of the hemagglutinin, known as the globular head domain and the process known as antigenic drift allows a virus to escape neutralization by mutating this part of hemagglutinin.
This genetic change, or shift, in the virus results in immunity to only specific strains of the influenza virus, requiring frequent reformulation and readministration of seasonal vaccines,”
Explains Peter Palese, PhD, Professor of Microbiology and Chair of the Department of Microbiology at Icahn School of Medicine at Mount Sinai, and co-author of the study. “Our chimeric HA vaccine, by contrast, is directed at the proximal part of the HA protein – the stalk domain – which has been shown to broadly neutralize diverse influenza virus strains in both animal models and humans,”
Dr. Palese emphasizes that an influenza vaccine that creates a strong and long-lasting immune response would not only end the need for annual reformulation but it would also eliminate the seasonal need for revaccination. “Revaccination annually is expensive and often inconvenient or even difficult to manage. Additionally, flu vaccine rates here in the United States represent an area where we see well-documented, persistent racial and ethnic disparities. A universal vaccine that lasted forever – or ideally at least 20 years – could certainly help relieve that imbalance,” Dr. Palese says.
The real prize associated with a universal, long-lasting vaccine goes beyond eliminating expenses and inconveniences, Dr. Palese adds. “If the majority of our population received an influenza vaccination, ideally with a high efficacy of 90%, then we would have herd immunity. We could potentially avoid an influenza pandemic of the same magnitude as we’re seeing now with COVID-19.”
- Krammer, F., Palese, P. Universal influenza virus vaccines: need for clinical trials. Nat Immunol 15, 3–5 (2014). https://doi.org/10.1038/ni.27613.3.
- Disease Burden of Influenza https://www.cdc.gov/flu/about/burden/index.html#:~:text=While%20the%20impact%20of%20flu,61%2C000%20deaths%20annually%20since%202010
- 1918 Pandemic (H1N1 virus) https://www.cdc.gov/flu/pandemic-resources/1918-pandemic-h1n1.html#:~:text=It%20is%20estimated%20that%20about,occurring%20in%20the%20United%20States.
- Past Seasons Vaccine Effectiveness Estimates https://www.cdc.gov/flu/vaccines-work/past-seasons-estimates.html